ThyroSeq® GC - the most comprehensive NGS test for thyroid nodules available
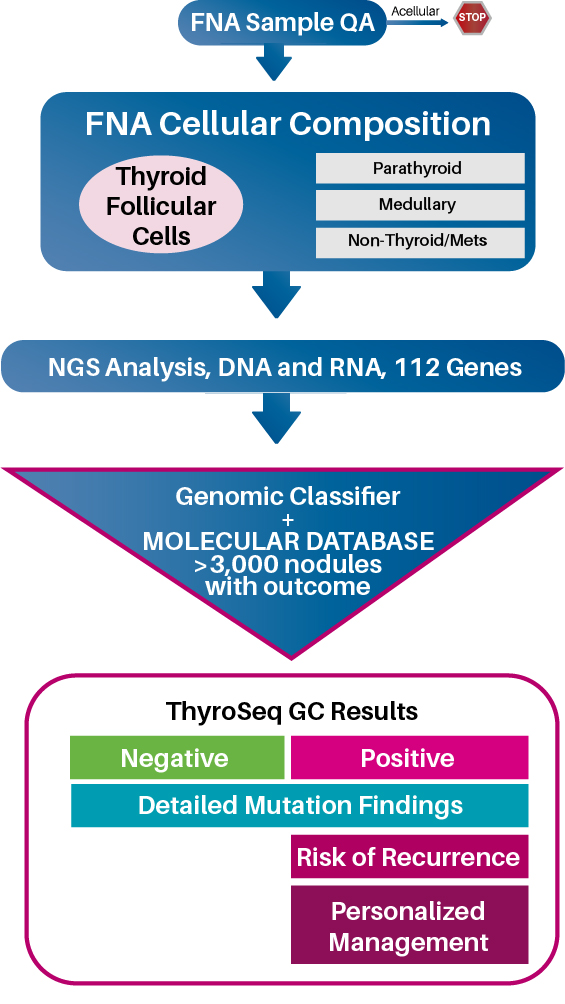
ThyroSeq® Genomic Classifier (GC) test utilizes next-generation sequencing to analyze DNA and RNA of 112 thyroid-related genes for four main classes of molecular alterations, including mutations, gene fusions, copy number alterations, and gene expression alterations
The results are processed by a unique Genomic Classifier (GC) and reported as Positive or Negative1
112 Thyroid-Related Genes
Including all expressed (BRAF, RAS) and not expressed (TERT) variants important for thyroid cancer
Including known and novel types of RET, NTRK, ALK, and other fusions
Critical for diagnosis of Hürthle cell nodules and follicular carcinomas
To diagnose medullary carcinoma, non-thyroidal nodules, and additional classes of thyroid cancer
In one test, ThyroSeq® Classifier:
-
Confidently rules out cancer and prevents unneeded surgeries in 61-67% of patients with indeterminate cytology nodules 2-8
-
Accurately detects all types of thyroid cancer (papillary, follicular, Hürthle cell, medullary) and non-thyroidal lesions (parathyroid, metastases to the thyroid) 1,2,7
-
Assesses risk of cancer recurrence
-
Identifies therapeutic targets for advanced cancer
ThyroSeq® Test Workflow
References:
1. Nikiforova MN, et al. Cancer. 2018. 2. Steward DL, et al. JAMA Oncol. 2018. 3. Chin PD, et al. Endocrin Pathol. 2020. 4. Jug R, et al. Cancer Cytopathol. 2020. 5. Guan H, et al. Thyroid. 2020. 6. Chen T, et al. Thyroid. 2020. 7. Schatz-Siemers N, et al. Diagn Cytopathol. 2019. 8. Desai D, et al. Cancer Cytopathol. 2020.
