Patient Management
ThyroSeq® test results refine cancer probability in thyroid nodules with indeterminate cytology, informing the most appropriate management of these patients.
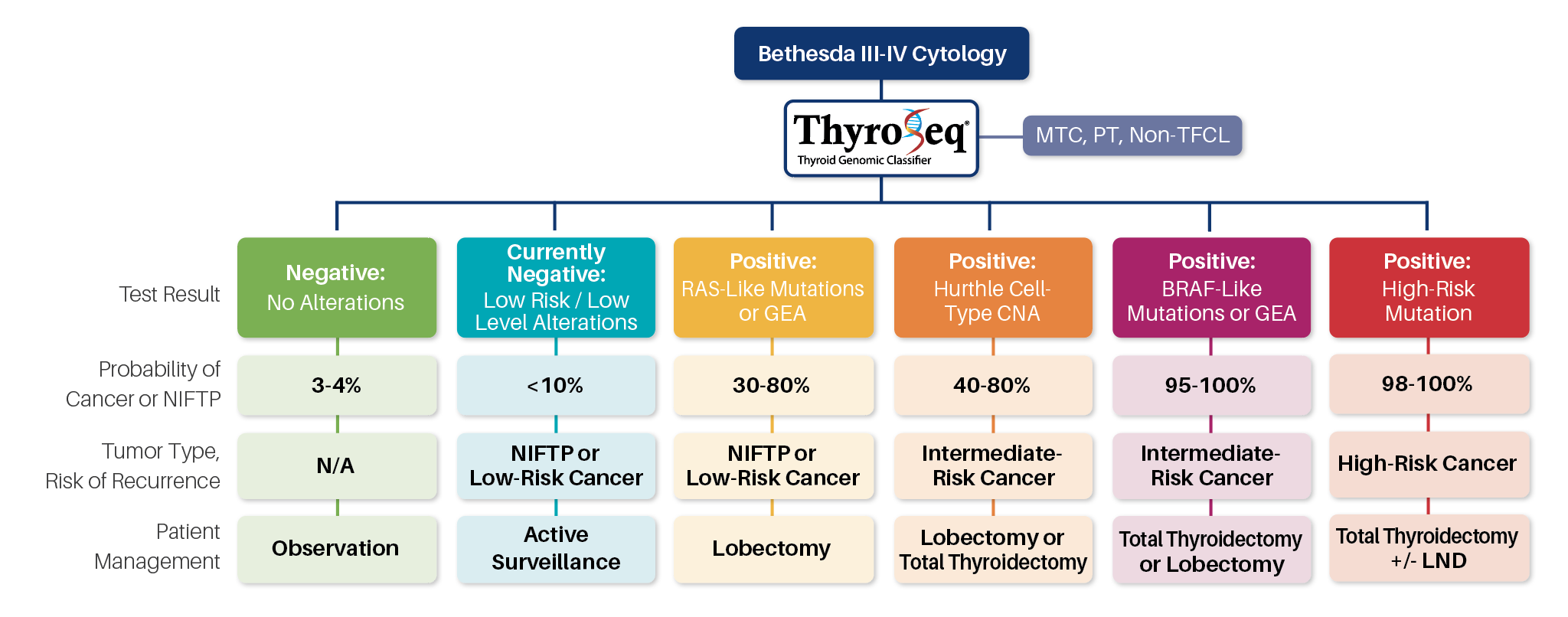
Abbreviations: MTC, medullary thyroid cancer; PT, parathyroid; Non-TFCL, non-thyroid follicular cell lesion; GEA, gene expression alterations; CNA, copy number alterations; LND, lymph node dissection.
NEGATIVE
RESULTS
CURRENTLY NEGATIVE
RESULTS
Therefore, in those clinical situations where the pretest probability of cancer in nodules with Bethesda III and IV cytology is <44%, negative ThyroSeq test results would confer the cancer probability of 5% or less, justifying observation in lieu of surgical management in appropriately selected cases. Because the probability of cancer in such nodules is comparable to benign FNA cytology, the management of patients may follow the recommendations for nodules with benign cytology, which, based on the 2015 ATA guidelines, should be determined based on ultrasound (US) pattern (Recommendation #23). In nodules with Bethesda V cytology and negative ThyroSeq result, the residual cancer risk of ~20% does not allow to avoid surgical management; thyroid lobectomy may be sufficient initial treatment for many of these patients.
Although at the time of sampling most of these nodules are benign, some of them may undergo clonal expansion and acquire additional mutations. In the absence of suspicious US features or other clinical risk factors, many of these patients are likely to benefit from active surveillance with repeat of clinical exam and potentially FNA and molecular testing in 1 year.
POSITIVE:
RAS-LIKE OR GEA
RESULTS
POSITIVE:
HÜRTHLE CELL-TYPE CNA
RESULTS
Many of these nodules may be managed by therapeutic lobectomy, which is currently recommended by the ATA guidelines for low-risk papillary and follicular carcinomas (Recommendation #35) and NIFTP.
POSITIVE:
BRAF-LIKE OR GEA
RESULTS
POSITIVE:
HIGH-RISK MUTATION
RESULTS
According to the ATA guidelines, BRAF-mutated
unifocal intrathyroidal carcinoma <1 cm in size has low risk for
recurrence and therefore may be treated with thyroid
lobectomy alone, whereas 1-4 cm BRAF-positive PTC is an
intermediate-risk tumor, where total thyroidectomy or
lobectomy should be considered based on clinical and US
findings.
Most of these patients would likely benefit from total thyroidectomy, with possible consideration for regional lymph node dissection if one of the mutations is BRAF V600E.
